Because of the central role of microtubules in mitosis, drugs that affect microtubule assembly are useful not only as experimental tools in cell biology but also in the treatment of cancer. As discussed later, this rapid turnover of microtubules is particularly critical for the remodeling of the cytoskeleton that occurs during mitosis. Growth of microtubules continues (more.)ĭynamic instability, described by Tim Mitchison and Marc Kirschner in 1984, results in the continual and rapid turnover of most microtubules, which have half-lives of only several minutes within the cell. Dynamic instability results from the hydrolysis of GTP bound to β-tubulin during or shortly after polymerization, which reduces its binding affinity for adjacent molecules. If this occurs, the GDP-bound tubulin will dissociate, resulting in rapid depolymerization and shrinkage of the microtubule.ĭynamic instability of microtubules. However, if the rate of polymerization slows, the GTP bound to tubulin at the plus end of the microtubule will be hydrolyzed to GDP. As long as new GTP-bound tubulin molecules are added more rapidly than GTP is hydrolyzed, the microtubule retains a GTP cap at its plus end and microtubule growth continues. Whether a microtubule grows or shrinks is determined by the rate of tubulin addition relative to the rate of GTP hydrolysis. In microtubules, GTP hydrolysis also results in the behavior known as dynamic instability, in which individual microtubules alternate between cycles of growth and shrinkage ( Figure 11.38). Like actin filaments (see Figure 11.4), microtubules undergo treadmilling, a dynamic behavior in which tubulin molecules bound to GDP are continually lost from the minus end and replaced by the addition of tubulin molecules bound to GTP to the plus end of the same microtubule. This GTP hydrolysis weakens the binding affinity of tubulin for adjacent molecules, thereby favoring depolymerization and resulting in the dynamic behavior of microtubules. In particular, the GTP bound to β-tubulin (though not that bound to α-tubulin) is hydrolyzed to GDP during or shortly after polymerization. Both α- and β- tubulin bind GTP, which functions analogously to the ATP bound to actin to regulate polymerization. Tubulin dimers can depolymerize as well as polymerize, and microtubules can undergo rapid cycles of assembly and disassembly. Dimers of α- and β-tubulin polymerize to form microtubules, which are composed of 13 protofilaments assembled around a hollow core. This polarity is an important consideration in determining the direction of movement along microtubules, just as the polarity of actin filaments defines the direction of myosin movement. Consequently, microtubules (like actin filaments) are polar structures with two distinct ends: a fast-growing plus end and a slow-growing minus end. The protofilaments, which are composed of head-to-tail arrays of tubulin dimers, are arranged in parallel. Tubulin dimers polymerize to form microtubules, which generally consist of 13 linear protofilaments assembled around a hollow core ( Figure 11.37). In addition, a third type of tubulin (γ-tubulin) is specifically localized to the centrosome, where it plays a critical role in initiating microtubule assembly (discussed shortly). Like actin, both α- and β-tubulin are encoded by small families of related genes. 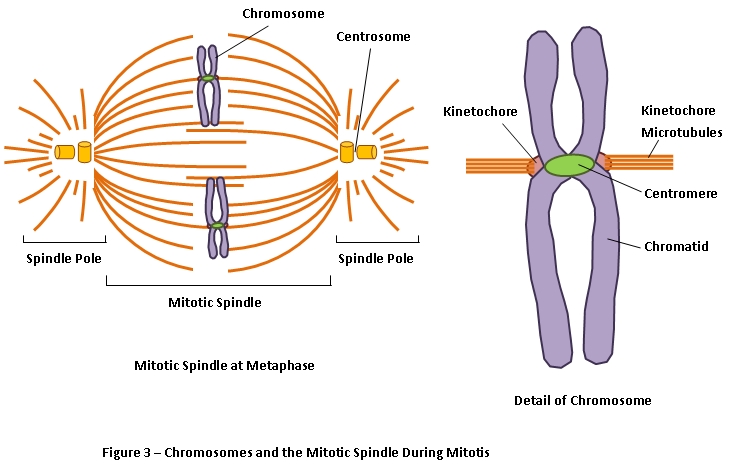
Tubulin is a dimer consisting of two closely related 55-kd polypeptides, α- tubulin and β-tubulin. In contrast to intermediate filaments, which are composed of a variety of different fibrous proteins, microtubules are composed of a single type of globular protein, called tubulin. Structure, Assembly, and Dynamic Instability of Microtubules 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
